Energy of hydrocarbon fuels converted directly from chemical form to electrical form
A C-H bond is formed by mutual sharing of an electron. Normally, when the bond breaks, energy is released in the form of heat. This invention aims to make the electron escape entirely, leaving an H+ ion and generating electricity directly instead of heat.
Invention (What is New)
By providing additional external catalytic energy (via electromagnetic waves), the shared electron in a C-H bond can escape, leaving behind H+ ions. The freed electron and H+ ion together form an electrical current source. The catalytic quantum of energy is re-used for subsequent cycles, making the process potentially self-sustaining.
Diagram & Description
Ionization energy for isolated H atom is 1310 kJ/mol, i.e. 2.1749 x 10-18 J per atom. Energy in one C-H bond is 0.3699 x 10-18 J. The extra 1.805 x 10-18 J must be provided externally as electromagnetic waves (~2.7 x 1015 Hz, wavelength ~1090 A).
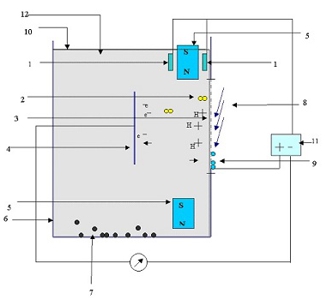
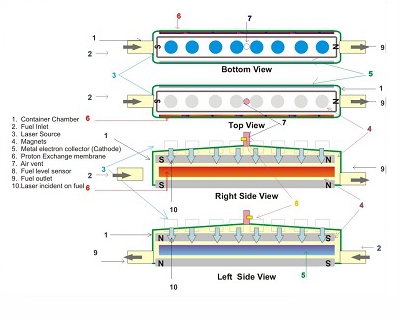
Working
The liquid hydrocarbon fuel is exposed to collimated electromagnetic waves in absence of air. Electrons escape from hydrogen atoms, becoming free electrons, while H atoms become H+ ions. A magnetic field directs electrons to a metal collector, and ions through a proton exchange membrane where they recombine with oxygen to form water. The electrodes act as charge collectors, driving current in an external circuit.
Calculation
| Energy per CH4 molecule | 1.4796 x 10-18 J |
| Energy per C-H bond in CH4 | 0.3699 x 10-18 J |
| Ionization energy per H atom | 2.1749 x 10-18 J |
| Extra catalytic quantum required | 1.805 x 10-18 J |
| Frequency of EM wave | ~2.7 x 1015 Hz |
| Wavelength of EM wave | ~1090 A |
Uses
This direct conversion process could replace internal combustion engines, which suffer ~60% energy losses, offering a cleaner and more efficient way to harness energy from hydrocarbon fuels.

